
Chemistry, 04.12.2019 06:31 jaedenevan062907
Which of the following statements explain why the van der waals equation must be used to describe real gases? x. interactions between gas molecules reduces the temperature of the gas in the sample y. the non-zero volumes of gas particles effectively decrease the amount of "empty space" between them z. the molecular attractions between particles of gas decreases the pressure exerted by the gas

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:30
What does x represent in the formula for the compound xcl4?
Answers: 2


Chemistry, 22.06.2019 11:40
Which type of precipitation would most likely form when the surface air temperature is slightly below freezing and the air temperature increases as you move upward away from the ground?
Answers: 2

You know the right answer?
Which of the following statements explain why the van der waals equation must be used to describe re...
Questions








Mathematics, 16.04.2021 19:50

Mathematics, 16.04.2021 19:50



History, 16.04.2021 19:50

Mathematics, 16.04.2021 19:50

Biology, 16.04.2021 19:50

Mathematics, 16.04.2021 19:50

Mathematics, 16.04.2021 19:50

Physics, 16.04.2021 19:50

History, 16.04.2021 19:50


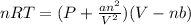 (1)
(1)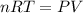 (2)
(2)

