Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2

Chemistry, 22.06.2019 08:00
Asap! will give brainiest when a heat wave strikes a region causing more people to run air-conditioning units, electrical demand increases. what needs to be done to meet this increased demand? raising the control rodslowering the control rodsremoving the control rods
Answers: 1

Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1

Chemistry, 23.06.2019 08:30
Explain how to convert from one unit to another in the metric system.
Answers: 3
You know the right answer?
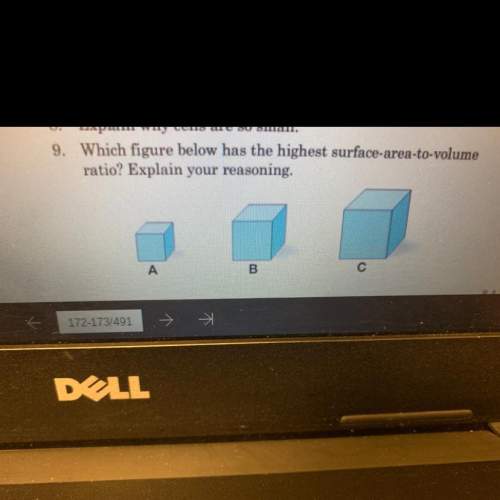
Ineed . give me an explanation. you so much.
...
...
Questions

Mathematics, 14.12.2020 17:20

Social Studies, 14.12.2020 17:20

Mathematics, 14.12.2020 17:20

Mathematics, 14.12.2020 17:20

Social Studies, 14.12.2020 17:20






Advanced Placement (AP), 14.12.2020 17:20



English, 14.12.2020 17:20


Social Studies, 14.12.2020 17:20



Mathematics, 14.12.2020 17:20

Arts, 14.12.2020 17:20