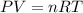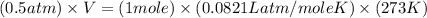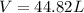Chemistry, 28.09.2019 08:00 lorip7799ov3qr0
The pressure of 1 mol of gas is decreased to 0.5 atm at 273 k. what happens to the molar volume of the gas under these conditions?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:00
Look at the reaction below: ca(hco3)2 --> caco3 + co2 + h2o first, balance the reaction. once balanced, use dimensional analysis or another method to find out how many moles of carbon dioxide will be produced if we start with 16.5 moles of calcium bicarbonate (calcium hydrogen carbonate). = mol of co2 number needs to be reported to three significant figures.
Answers: 1

Chemistry, 21.06.2019 20:00
Smog is the term used to describe the combination of fog and smoke
Answers: 1


Chemistry, 22.06.2019 21:00
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1
You know the right answer?
The pressure of 1 mol of gas is decreased to 0.5 atm at 273 k. what happens to the molar volume of t...
Questions






Mathematics, 01.02.2020 00:43

Mathematics, 01.02.2020 00:43

English, 01.02.2020 00:43


Mathematics, 01.02.2020 00:43



Mathematics, 01.02.2020 00:43


Health, 01.02.2020 00:43


Chemistry, 01.02.2020 00:44



Mathematics, 01.02.2020 00:44