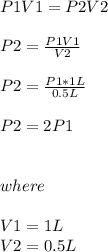Chemistry, 03.12.2019 23:31 lilybrok04
One liter of a gas is in a sealed chamber containing a moveable piston. if the piston is moved so that the volume of the gas is compressed to a volume of one-half liter, what will happen to the pressure on the gas? (assume the temperature is constant and no gas particles are lost.)
a)the pressure will be twice the original value.
b)the pressure will be half of the original value.
c)it would be impossible to move the piston since gases are not compressible.
d)the pressure will remain the same.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:00
According to the tide table below what time of day will the highest tide occur? (2 pt) the highest tide will occur at
Answers: 1

Chemistry, 23.06.2019 09:00
Agust of wind blowing east pushes against a ball. when will the wind do work on the ball? when the ball moves to the east when the ball moves to the north when the ball stays in one place when the ball moves north or south
Answers: 1

Chemistry, 23.06.2019 15:30
Among these processes, which is the slowest chemical reaction? a. digesting food b. boiling an egg c. tarnishing of silver d. melting of a glacier
Answers: 2

Chemistry, 23.06.2019 15:40
The table below shows the freezing points of four substances. substance freezing point (°c) benzene 5.5 water 0 butane –138 nitrogen –210 the substances are placed in separate containers at room temperature, and each container is gradually cooled. which of these substances will solidify before the temperature reaches 0°c? benzene water butane nitrogen
Answers: 2
You know the right answer?
One liter of a gas is in a sealed chamber containing a moveable piston. if the piston is moved so th...
Questions

Mathematics, 06.05.2020 09:00

Biology, 06.05.2020 09:00

Mathematics, 06.05.2020 09:00

Mathematics, 06.05.2020 09:00

History, 06.05.2020 09:00

Mathematics, 06.05.2020 09:00

Mathematics, 06.05.2020 09:00


Mathematics, 06.05.2020 09:00



Mathematics, 06.05.2020 09:00


Mathematics, 06.05.2020 09:00


Mathematics, 06.05.2020 09:00

Mathematics, 06.05.2020 09:00