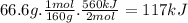Achemist measures the energy change ? h during the following reaction:
2fe2o3(s) ? 4fe...

Achemist measures the energy change ? h during the following reaction:
2fe2o3(s) ? 4feo(s) + o2(g) =? h560.kj
this reaction is.
(a) endothermic
(b) exothermic
suppose 66.6 g of fe2o3 react. will any heat be released or absorbed? |
(a) yes, absorbed
(b) yes released.
(c) no

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 13:30
Why are gamma rays not affected by a magnet as they pass over it? gamma rays are composed of only energy. gamma rays do not have enough mass to be affected. gamma rays do not have the right electrical charge to be affected. gamma rays move too fast for anything to affect their pathway.
Answers: 1

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3


Chemistry, 22.06.2019 07:40
22. a flask containing 450 ml of 0.50 m h2so4 was accidentally knocked to the floor. how many grams of nahco, do you need to put on the spill to neutralize the acid according to the following equation: h2so4(aq)+2 nahcos(aq) na,so(aq) +2 h20()+2 co2(g) d) 38 g a) 2.3 g b) 9.5 g c) 19 g
Answers: 1
You know the right answer?
Questions



Physics, 27.02.2020 01:27



History, 27.02.2020 01:27




Computers and Technology, 27.02.2020 01:27



Mathematics, 27.02.2020 01:28

Computers and Technology, 27.02.2020 01:28




History, 27.02.2020 01:28