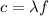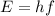Chemistry, 03.12.2019 06:31 genyjoannerubiera
Fireworks that contain metallic salts such as sodium, strontium, and barium can generate bright colors. a technician investigates what colors are produced by the metallic salts by performing flame tests. during a flame test, a metallic salt is heated in the flame of a gas burner. each metallic salt emits a characteristic colored light in the flame. 3. state how bright-line spectra viewed through a spectroscope can be used to identify the metal ions in the salts used in the flame tests. 4. explain, in terms of electrons, how a strontium salt emits colored light. 5. explain why the electron configuration of 2-7-1-1 represents a sodium atom in an excited state.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:00
Review the branily terms and services guides well u know what i never did so go have a nice ice cream sunday
Answers: 1

Chemistry, 22.06.2019 21:00
In the experiment you asked to react hydrochloric acid and with sodium hydroxide. when measuring the volume of the reactants, which instrument would give the greatest precision.
Answers: 3


You know the right answer?
Fireworks that contain metallic salts such as sodium, strontium, and barium can generate bright colo...
Questions

Biology, 07.11.2020 04:30

Mathematics, 07.11.2020 04:30



Law, 07.11.2020 04:30

Mathematics, 07.11.2020 04:30


History, 07.11.2020 04:30




Mathematics, 07.11.2020 04:30

Mathematics, 07.11.2020 04:30


History, 07.11.2020 04:30



Chemistry, 07.11.2020 04:30


Chemistry, 07.11.2020 04:30