
Two flasks of equal volume and at the same temperature contain different gases. one flask contains 5.0 g of o2, and the other flask contains 5.0 g of h2. is each of the following statements true or false? explain.
a) true. because the gases have the same volumes, they must have the same number of molecules.
b) false. because the molar mass of o2 is greater than the molar mass of h2, 5.0g of o2 will contain fewer molecules than 5.0 g of h2.
c)false. depending on the pressure each flask may contain different numbers of molecules.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Substance x has a fixed volume, and the attraction between its particles is strong .substance y had widely spread out particles and can be compressed what can most likely be concluded about these substances
Answers: 2

Chemistry, 22.06.2019 00:00
Explain which group an element with the electron configuration 1s2 2s2 2p6 3s2 3p6 3d1 4s2 belongs to.
Answers: 3


You know the right answer?
Two flasks of equal volume and at the same temperature contain different gases. one flask contains 5...
Questions




History, 09.08.2021 20:50

History, 09.08.2021 20:50

World Languages, 09.08.2021 20:50





Health, 09.08.2021 20:50



Computers and Technology, 09.08.2021 20:50

Mathematics, 09.08.2021 20:50

Mathematics, 09.08.2021 20:50

Social Studies, 09.08.2021 20:50


Mathematics, 09.08.2021 20:50

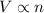
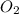 =
= 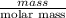
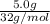
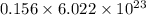
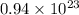 molecules
molecules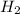 =
= 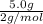
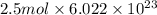
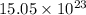 molecules
molecules![O_{2} will contain same molecules as 5.0 g of [tex]H_{2}](/tpl/images/0400/5815/5c250.png) is not true.
is not true.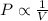 (at constant temperature and number of moles)
(at constant temperature and number of moles)

