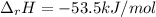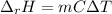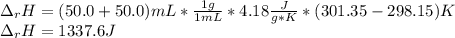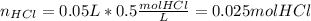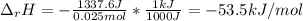Chemistry, 03.12.2019 04:31 dhananjaynagarkar
When 50.0 ml of 0.500 m hcl at 25.0°c is added to 50.0 ml of 0.500 m naoh at 25.0°c in a coffee cup calorimeter, the temperature of the mixture rises to 28.2°c.
what is the enthalpy of reaction per mole of acid?
assume the mixture has a specific heat capacity of 4.18 j/(g ? k) and that the densities of the reactant solutions are both 1.00 g/ml.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 10:00
Nonpoint source pollution is difficult to control because it
Answers: 2

Chemistry, 22.06.2019 15:00
Which theory was contradicted by experiments with the photoelectric effect? light spreads out after it passes through a small opening. as soon as light strikes metal, electrons will be ejected. visible light, regardless of color, will cause the ejection of electrons when striking metal. the kinetic energy of ejected electrons depends on the frequency of light that strikes the metal.
Answers: 2

Chemistry, 22.06.2019 15:30
The reactions of photosynthesis occur in the of plant cell? a.mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1
You know the right answer?
When 50.0 ml of 0.500 m hcl at 25.0°c is added to 50.0 ml of 0.500 m naoh at 25.0°c in a coffee cup...
Questions


Computers and Technology, 14.12.2019 03:31