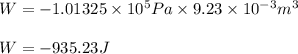Chemistry, 03.12.2019 04:31 jasmine2919
Automobile airbags contain solid sodium azide, nan 3 , that reacts to produce nitrogen gas when heated, thus inflating the bag. 2 nan 3 ( s ) ⟶ 2 na ( s ) + 3 n 2 ( g ) calculate the value of work, w , for the system if 16.5 g nan 3 reacts completely at 1.00 atm and 22 ∘ c.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:30
The chart shows the bid provided by four contractors to complete a job. which contractor is the most cost-effective?
Answers: 3

Chemistry, 22.06.2019 12:20
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3

Chemistry, 23.06.2019 08:00
Can anyone answer these questions? ? i need it before 1: 00pm today
Answers: 1

Chemistry, 23.06.2019 10:30
If a 20.0ml test tube measures 15.0cm, what is the length in meters?
Answers: 1
You know the right answer?
Automobile airbags contain solid sodium azide, nan 3 , that reacts to produce nitrogen gas when heat...
Questions

Mathematics, 10.12.2021 17:20

Mathematics, 10.12.2021 17:20


Computers and Technology, 10.12.2021 17:20


Mathematics, 10.12.2021 17:20

Mathematics, 10.12.2021 17:20



English, 10.12.2021 17:20

Social Studies, 10.12.2021 17:20

Biology, 10.12.2021 17:20

Biology, 10.12.2021 17:20


Physics, 10.12.2021 17:20

Social Studies, 10.12.2021 17:20

Mathematics, 10.12.2021 17:20

Physics, 10.12.2021 17:20

History, 10.12.2021 17:20

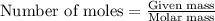
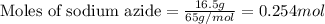
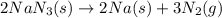
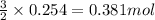 of nitrogen gas
of nitrogen gas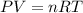
![22^oC=[22+273]K=295K](/tpl/images/0400/4731/7919f.png)
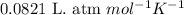
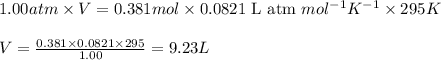
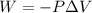
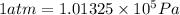 (Conversion factor: 1 atm = 101325 Pa)
(Conversion factor: 1 atm = 101325 Pa)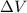 = change in volume =
= change in volume = 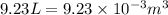 (Conversion factor:
(Conversion factor: 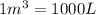 )
)