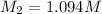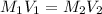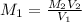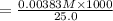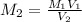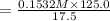Chemistry, 29.11.2019 06:31 sarahjdeering
A17.55 ml solution of potassium nitrate (kno3) was diluted to 125.0 ml, and 25.00 ml of this solution was then diluted to 1.000 × 103 ml. the concentration of the final solution is 0.00383 m. calculate the concentration of the original solution.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 15:00
Which element in the third period would you expect to have the larger atomic radius, sodium (na) or sulfur (s)? a. sodium, because it has a higher effective nuclear charge attracting electrons in fewer energy levels. b. sodium, because it has fewer protons attracting electrons in the same energy levels. c. sulfur, because it has more protons attracting electrons in more energy levels. d. sulfur, because it has a higher effective nuclear charge attracting electrons in the same energy levels.
Answers: 2

Chemistry, 22.06.2019 03:30
What is the relationship of air masses and the temperature of oceans?
Answers: 1

Chemistry, 22.06.2019 06:00
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 13.0 mol cesium fluoride with 12.5 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1

Chemistry, 22.06.2019 09:20
Explain that newton first law,second law and third law of motion?
Answers: 2
You know the right answer?
A17.55 ml solution of potassium nitrate (kno3) was diluted to 125.0 ml, and 25.00 ml of this solutio...
Questions

Mathematics, 04.04.2021 20:00

History, 04.04.2021 20:00


History, 04.04.2021 20:00

Physics, 04.04.2021 20:00

Social Studies, 04.04.2021 20:00

Mathematics, 04.04.2021 20:00

Mathematics, 04.04.2021 20:00

Mathematics, 04.04.2021 20:00










History, 04.04.2021 20:00