
Aspirin (acetylsalicylic acid, c9h8o4) is a weak monoprotic acid. to determine its acid-dissociation constant, a student dissolved 2.00 g of aspirin in 0.600 l of water and measured the ph. what was the ka value calculated by the student if theph of the solution was 2.62? a 0.100 m solution of ethylamine (c2h5nh2) has a ph of 11.87. calculate the kb for ethylamine.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:20
Use the gizmo to find the concentration of the mystery ch3cooh. use the titrant and indicator shown below perform the titration. what is the titrant volume? titrant analyte indicator titrant volume analyte concentration naoh ch3cooh phenophthalein select one: a. 20.0 ml b. 27.0 ml c. 30.0 ml d. 24.0 ml
Answers: 2


Chemistry, 22.06.2019 14:00
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1

Chemistry, 22.06.2019 22:10
Determine the ph of 0.10 m nh3 solution. nh3 is a weak base with a kb equal to 1.8 x 10-5 round answer to nearest whole number.
Answers: 1
You know the right answer?
Aspirin (acetylsalicylic acid, c9h8o4) is a weak monoprotic acid. to determine its acid-dissociation...
Questions


Business, 31.03.2021 14:00


Mathematics, 31.03.2021 14:00


Mathematics, 31.03.2021 14:00


Mathematics, 31.03.2021 14:00




Biology, 31.03.2021 14:00

Mathematics, 31.03.2021 14:00

Chemistry, 31.03.2021 14:00


Mathematics, 31.03.2021 14:00

English, 31.03.2021 14:00


English, 31.03.2021 14:00

Computers and Technology, 31.03.2021 14:00

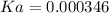
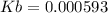
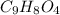 = 180.16 g/mol
= 180.16 g/mol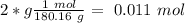
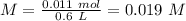
![Ka=\frac{[X][X]}{[0.019-X]}=\frac{[X]^2}{[0.019-X]}](/tpl/images/0395/6760/646b6.png)
![pH=-Log[H^+]](/tpl/images/0395/6760/3ca39.png)
![[H^+]=10^-^p^H](/tpl/images/0395/6760/16952.png)
![[H^+]=10^-^2^.^6^2=0.00240](/tpl/images/0395/6760/b62ea.png)
![Ka=\frac{[0.00240]^2}{[0.019-0.00240]}=0.000346](/tpl/images/0395/6760/6c4c9.png) .
.![Kb=\frac{[X][X]}{[0.1-X]}=\frac{[X]^2}{[0.1-X]}](/tpl/images/0395/6760/c1bde.png)
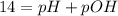
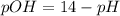
![pOH=-Log[OH^-]](/tpl/images/0395/6760/626fd.png)
![[OH^-]=10^-^p^O^H](/tpl/images/0395/6760/86a8e.png)
![[OH^-]=10^-^2^.^1^3=0.00741](/tpl/images/0395/6760/49ab9.png)
![Kb=\frac{[0.00741]^2}{[0.1-0.00741]}=0.000593](/tpl/images/0395/6760/bbeaa.png)


