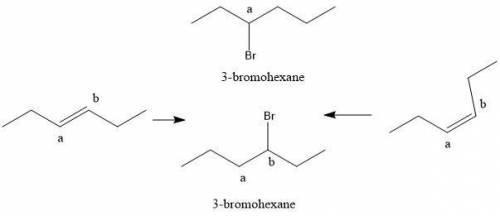
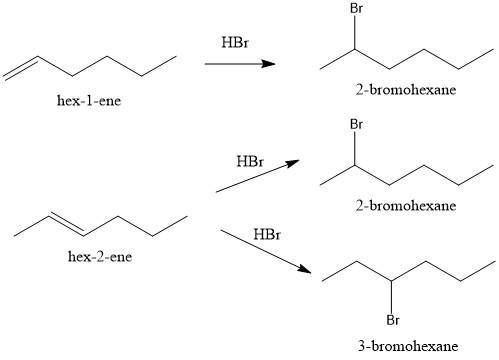
Compounds x and y are stereoisomers having the formula c6h12. both x and y react with one molar equivalent of hydrogen in the presence of a palladium catalyst to form hexane, and they each react with hbr to give a single bromoalkane product. draw structural formulas for both x and y.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 11:40
Which type of precipitation would most likely form when the surface air temperature is slightly below freezing and the air temperature increases as you move upward away from the ground?
Answers: 2

Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1

Chemistry, 23.06.2019 01:00
Chromium(iii) sulfate is a transition metal compound containing the metal chromium and the polyatomic ion sulfate. the oxidation state of chromium in this compound is , and the chemical formula of the compound is ( ) . reset next
Answers: 3
You know the right answer?
Compounds x and y are stereoisomers having the formula c6h12. both x and y react with one molar equi...
Questions




English, 21.02.2020 00:11





Business, 21.02.2020 00:12




Arts, 21.02.2020 00:12

Physics, 21.02.2020 00:13



Physics, 21.02.2020 00:13


Mathematics, 21.02.2020 00:13