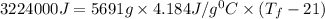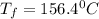The balanced combustion reaction for c 6 h 6 is 2 c 6 h 6 ( l ) 15 o 2 ( g ) ⟶ 12 co 2 ( g ) 6 h 2 o ( l ) 6542 kj if 7.700 g c 6 h 6 is burned and the heat produced from the burning is added to 5691 g of water at 21 ∘ c, what is the final temperature of the water

Answers: 2
Another question on Chemistry



Chemistry, 23.06.2019 00:20
How many lone pairs of electrons are on the central atom of no3- and what is the molecular shape? one, trigonal planar zero, trigonal pyramidal zero, trigonal planar one, tetrahedral one, trigonal pyramidal
Answers: 1

Chemistry, 23.06.2019 14:00
If the molar mass of the compound is 96.69 g/mol, what is the molecular formula of the compound?
Answers: 1
You know the right answer?
The balanced combustion reaction for c 6 h 6 is 2 c 6 h 6 ( l ) 15 o 2 ( g ) ⟶ 12 co 2 ( g ) 6 h 2 o...
Questions

English, 07.11.2020 19:50

Mathematics, 07.11.2020 19:50



Computers and Technology, 07.11.2020 19:50

Mathematics, 07.11.2020 19:50


Mathematics, 07.11.2020 19:50




History, 07.11.2020 19:50

Physics, 07.11.2020 19:50



Mathematics, 07.11.2020 19:50

English, 07.11.2020 20:00


Arts, 07.11.2020 20:00

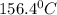
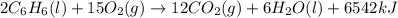
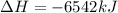
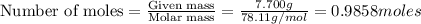
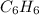 releases = 6542 kJ of heat
releases = 6542 kJ of heat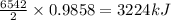 of heat
of heat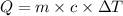
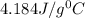
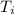 = 21.0°C
= 21.0°C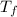 = ?
= ?