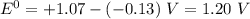Chemistry, 29.11.2019 00:31 savyblue1724707
Use the standard half-cell potentials listed below to calculate the standard cell potential for the following reaction occurring in an electrochemical cell at 25°c. (the equation is balanced.) pb(s) + br2(l) → pb2+(aq) + 2br(aq) pb2+(aq) + 2 e → pb(s) e° = -0.13 v br2(l) + 2 e → 2 br(aq) e° = +1.07 v

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:30
You have 125g of a certain seasoning and are told that it contains 76.0 g of salt what is the percentage of salt by mass in this seasoning
Answers: 1

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

Chemistry, 22.06.2019 05:00
If the density of water is 1.0 g/cm3, which of these materials would float in water, based on their densities? check all that apply. aluminum cork iron lead wax
Answers: 1

You know the right answer?
Use the standard half-cell potentials listed below to calculate the standard cell potential for the...
Questions

History, 01.01.2021 02:50



Health, 01.01.2021 02:50

Health, 01.01.2021 02:50

Mathematics, 01.01.2021 02:50













Mathematics, 01.01.2021 03:00

Mathematics, 01.01.2021 03:00

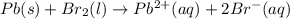
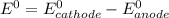
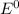 are standard reduction potentials.
are standard reduction potentials.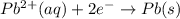
![E^0_{[Pb^{2+}/Pb]}= -0.13\ V](/tpl/images/0395/3322/82712.png)
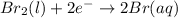
![E^0_{[Br_2/Br^{-}]}=+1.07\ V](/tpl/images/0395/3322/f8f7e.png)
![E^0=E^0_{[Br_2/Br^{-}]}- E^0_{[Pb^{2+}/Pb]}](/tpl/images/0395/3322/fba3a.png)