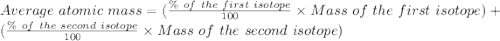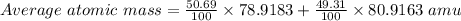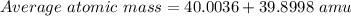Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Anurse practitioner prepares an injection of promethazine, an antihistamine used to treat allergic rhinitis. if the stock bottle is labeled 25 mg/ml and the order is a dose of 11.0 mg , how many milliliters will the nurse draw up in the syringe?
Answers: 3

Chemistry, 22.06.2019 04:50
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 05:30
Match the following vocabulary terms to their definitions. 1. amount of energy required to change 1 gram of material from the solid to the liquid state at its melting point 2. a measure of the kinetic energy of the particles of a substance 3. the amount of heat energy required to raise the temperature of 1 gram of liquid water from 14.5°c to 15.5°c 4. amount of energy required to change 1 gram of material from the liquid to the gaseous state at its boiling point 5. the amount of energy required to change 1 gram of a substance 1°c a. temperature b. latent heat of vaporization c. latent heat of fusion d. calorie e. specific heat
Answers: 1

Chemistry, 22.06.2019 19:30
Acetylene gas c2h2 undergoes combustion to produce carbon dioxide and water vapor how many grams of water are produced by the same amount of c2h2?
Answers: 2
You know the right answer?
Bromine has two isotopes 79br and 81br, whose masses (78.9183 and 80.9163 amu) and abundances (50.69...
Questions

English, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01

Mathematics, 18.09.2020 05:01