
Chemistry, 28.11.2019 22:31 leannadoughty06
A3.452 g sample containing an unknown amount of a ce(iv) salt is dissolved in 250.0-ml of 1 m h2so4. a 25.00 ml aliquot is analyzed by adding ki and titrating the i3– that forms with s2o32–. the end point was reached following the addition of 13.02 ml of 0.03428 m na2s2o3. calculate the weight percent of ce4 in the sample. when should the indicator be added to this titration? (at the beginning or just before the end point? )

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Plz me get these answer dubble cheak ur answer plz ppl i need it right
Answers: 2


Chemistry, 22.06.2019 19:00
Sum of brother and sisters age is 26. four times the brothers age is subtracted from three times the sisters age, the difference is 8. what are the ages of the brother and sister?
Answers: 1

Chemistry, 23.06.2019 01:30
Which statement accurately represents the arrangement of electrons in bohr’s atomic model?
Answers: 2
You know the right answer?
A3.452 g sample containing an unknown amount of a ce(iv) salt is dissolved in 250.0-ml of 1 m h2so4....
Questions

English, 27.03.2021 01:00



Mathematics, 27.03.2021 01:00

Physics, 27.03.2021 01:00

Mathematics, 27.03.2021 01:00

French, 27.03.2021 01:00

Chemistry, 27.03.2021 01:00

Mathematics, 27.03.2021 01:00






Mathematics, 27.03.2021 01:00

English, 27.03.2021 01:00


History, 27.03.2021 01:00

English, 27.03.2021 01:00

History, 27.03.2021 01:00

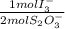 = 2,2315x10⁻⁴ moles of I₃⁻
= 2,2315x10⁻⁴ moles of I₃⁻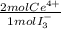 = 4,463x10⁻⁴ moles of Ce(IV). These moles are:
= 4,463x10⁻⁴ moles of Ce(IV). These moles are: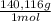 = 0,0625 g of Ce(IV)
= 0,0625 g of Ce(IV)

