Chemistry, 28.11.2019 03:31 jaylabeatty44
Use the standard half-cell potentials listed below to calculate the standard cell potential for the following reaction occurring in an electrochemical cell at 25�c. (the equation is balanced.)
3 cl2(g) + 2 fe(s) --> 6 cl-(aq) + 2 fe3+(aq)
cl2(g) + 2 e- --> 2 cl-(aq); e� = +1.36 v
fe3+(aq) + 3 e- --> fe(s); e� = -0.04 v
+1.32 v
-1.32 v
-1.40 v
+1.40 v
+4.16 v

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:30
Margaret wants to make an orange flavored drink by stirring powdered drink mix into a glass of water. she doesn't like drinks that have small clumps of powdered solid in them, so she wants the drink to be a perfect solution. what factors should margaret not consider when deciding how much powder to add to her glass of water?
Answers: 3

Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 14:00
How many absorptions would you expect to observe in the 13c nmr spectra of the following molecules? a) 3-chloropentane b) cis-4-methyl-2-pentene
Answers: 2

Chemistry, 22.06.2019 15:30
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks.energy was destroyed inside the blocks.energy was absorbed into the blocks from outside the system.energy was transferred from the warmer block to the cooler block.
Answers: 2
You know the right answer?
Use the standard half-cell potentials listed below to calculate the standard cell potential for the...
Questions


Chemistry, 26.06.2019 08:20


Mathematics, 26.06.2019 08:20


Mathematics, 26.06.2019 08:20

Biology, 26.06.2019 08:20

Mathematics, 26.06.2019 08:20


Mathematics, 26.06.2019 08:20


Mathematics, 26.06.2019 08:20


Mathematics, 26.06.2019 08:20


English, 26.06.2019 08:20




Physics, 26.06.2019 08:20

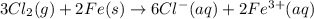
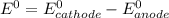
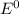 are standard reduction potentials.
are standard reduction potentials.
![E^0_{[Fe^{3+}/Fe]}=-0.04V](/tpl/images/0394/1963/07a45.png)
![E^0_{[Cl_2/Cl^-]}=+1.36V](/tpl/images/0394/1963/05c05.png)
![E^0=E^0_{[Cl_2/Cl^-]}- E^0_{[Fe^{3+}/Fe]}](/tpl/images/0394/1963/d357e.png)