
Chemistry, 28.11.2019 02:31 amanquen35
Find the equilibrium partial pressures of a and b for each of the following different values of kp.? consider the following reaction: a(g) = 2b(g)find the equilibrium partial pressures of a and b for each of the following different values of kp. assume that the initial partial pressure of b in each case is 1.0 atm and that the initial partial pressure of a is 0.0 atm. make any appropriate simplifying assumptions. kp = 1.4? kp = 2.0 * 10^-4? kp = 2.0 * 10^5?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:10
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3

Chemistry, 22.06.2019 06:00
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 13.0 mol cesium fluoride with 12.5 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1

Chemistry, 22.06.2019 08:30
Agroup of students is studying convection current. they fill two identical balloons with the same amount of helium. one balloon is placed in a freezer and the other is in an area with warm air. after 10 minutes, the balloon are released from a height of 1 meter. which of the following to the students most likely observe? a) the warm balloon expands and rises. the cold balloon shrinks and sinks b) the balloon both rise. the cold balloon is larger than the warm balloon c) the cold balloon expands and rises. the warm balloon shrinks and sinks d) the balloon rise at the same rate. both balloons are the same size
Answers: 1

Chemistry, 22.06.2019 12:00
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1
You know the right answer?
Find the equilibrium partial pressures of a and b for each of the following different values of kp.?...
Questions

Mathematics, 31.08.2021 03:50



Mathematics, 31.08.2021 03:50

Mathematics, 31.08.2021 03:50

Mathematics, 31.08.2021 03:50


Mathematics, 31.08.2021 03:50



History, 31.08.2021 03:50


History, 31.08.2021 03:50


Biology, 31.08.2021 03:50


Arts, 31.08.2021 03:50

English, 31.08.2021 03:50

Mathematics, 31.08.2021 03:50

Mathematics, 31.08.2021 03:50

![P_{[A] = 0,22](/tpl/images/0394/1164/9afd7.png) ,
, ![P_{[B] = 0,56atm](/tpl/images/0394/1164/02e2b.png)
![P_{[A] = 0,495](/tpl/images/0394/1164/54750.png) ,
, ![P_{[B] = 0,01atm](/tpl/images/0394/1164/da9a1.png)
![P_{[A] = 5x10^{-6}](/tpl/images/0394/1164/9be03.png) ,
, ![P_{[B] = 0,99999atm](/tpl/images/0394/1164/07800.png)
![kp = \frac{P_{[B]}^2}{P_{[A]}}](/tpl/images/0394/1164/0f2b8.png)
![P_{[A] = 0,0atm + X](/tpl/images/0394/1164/62221.png)
![P_{[B] = 1,0atm - 2X](/tpl/images/0394/1164/dd6ae.png)
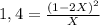
![P_{[B] = 1,0atm - 0,44atm = 0,56atm](/tpl/images/0394/1164/4f71b.png)
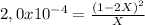
![P_{[A] = 0,495atm](/tpl/images/0394/1164/e1edd.png)
![P_{[B] = 1,0atm - 0,99atm = 0,01atm](/tpl/images/0394/1164/aeba4.png)
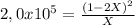
![P_{[B] = 1,0atm - 0,00001atm = 0,99999atm](/tpl/images/0394/1164/ca009.png)


