Given the standard enthalpy changes for the following two reactions:
(1) n2(g) + 2o2(g)2no2(...


Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3

Chemistry, 23.06.2019 01:00
The primary products of complete combustion of fossil fuels are a. carbon dioxide and water b. methane and water c. carbon monoxide and water d. carbon dioxide and carbon monoxide
Answers: 1

Chemistry, 23.06.2019 01:10
Volume is a measurement of how fast particles of a substance are moving
Answers: 3
You know the right answer?
Questions





Social Studies, 27.08.2019 14:00

Biology, 27.08.2019 14:00

SAT, 27.08.2019 14:00


History, 27.08.2019 14:00

Mathematics, 27.08.2019 14:00

Mathematics, 27.08.2019 14:00


Physics, 27.08.2019 14:00

Mathematics, 27.08.2019 14:00


Mathematics, 27.08.2019 14:00

Computers and Technology, 27.08.2019 14:00

Social Studies, 27.08.2019 14:00

History, 27.08.2019 14:00

 ...... ΔH° = 66.4 kJ
...... ΔH° = 66.4 kJ
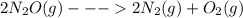 ......ΔH° = -164.2 kJ
......ΔH° = -164.2 kJ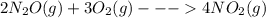 ......ΔH° = _________?
......ΔH° = _________?

