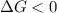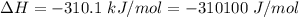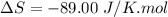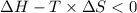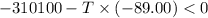Chemistry, 26.11.2019 03:31 Rileyb101207
Areaction will be spontaneous only at low temperatures if both δh and δs are negative. for a reaction in which δh = −310.1 kj/mol and δs = −89.00 j/k · mol, determine the temperature (in °c) below which the reaction is spontaneous.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:00
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2


Chemistry, 22.06.2019 14:00
How does the presence of oxygen affect the chemical pathways used to extract energy from glucose?
Answers: 3

Chemistry, 22.06.2019 21:00
In the experiment you asked to react hydrochloric acid and with sodium hydroxide. when measuring the volume of the reactants, which instrument would give the greatest precision.
Answers: 3
You know the right answer?
Areaction will be spontaneous only at low temperatures if both δh and δs are negative. for a reactio...
Questions

Social Studies, 11.09.2020 18:01

Mathematics, 11.09.2020 18:01

Biology, 11.09.2020 18:01

Mathematics, 11.09.2020 18:01

English, 11.09.2020 18:01

Geography, 11.09.2020 18:01

Social Studies, 11.09.2020 18:01

Mathematics, 11.09.2020 18:01

Mathematics, 11.09.2020 18:01

Mathematics, 11.09.2020 18:01

Mathematics, 11.09.2020 18:01

Mathematics, 11.09.2020 18:01

English, 11.09.2020 18:01

Mathematics, 11.09.2020 18:01

Biology, 11.09.2020 18:01

Mathematics, 11.09.2020 18:01

Mathematics, 11.09.2020 18:01

Social Studies, 11.09.2020 18:01

Mathematics, 11.09.2020 18:01

Mathematics, 11.09.2020 18:01

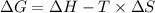
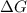 is the change in the Gibbs free energy.
is the change in the Gibbs free energy.
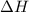 is the enthalpy change of the reaction.
is the enthalpy change of the reaction.
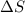 is the change in entropy.
is the change in entropy.