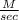The table shows the concentration of a reactant in the reaction mixture over a period of time.
...

Chemistry, 26.11.2019 03:31 michaela134
The table shows the concentration of a reactant in the reaction mixture over a period of time.
reactant concentrationtime concentration
0 s 1.8 m
210 s 1.2 m
450 s 0.8 m
580 s 0.6 m
720 s 0.4 m
what is the average rate of the reaction over the first 580 seconds?
1.6 × 10−3
1.9 × 10−3
2.0 × 10−3
2.2 × 10−3

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 20:00
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2

Chemistry, 22.06.2019 21:30
Achemical reaction is done in the setup shown, resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 1
You know the right answer?
Questions



Geography, 14.12.2020 08:40

Mathematics, 14.12.2020 08:40



Physics, 14.12.2020 08:40

Arts, 14.12.2020 08:40

Chemistry, 14.12.2020 08:40




English, 14.12.2020 08:40

History, 14.12.2020 08:40




Geography, 14.12.2020 08:40


Mathematics, 14.12.2020 08:40

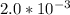
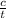 =
= 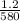 ≅
≅