
A0.327-g sample of azulene (c10h8) is burned in a bomb calorimeter and the temperature increases from 25.20 °c to 27.60 °c. the calorimeter contains 1.17×103 g of water and the bomb has a heat capacity of 786 j/°c. based on this experiment, calculate δe for the combustion reaction per mole of azulene burned (kj/mol). c13h24o4(s) + 17 o2(g) 13 co2(g) + 12 h2o(l) e = kj/mol.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:30
Calculate the expected ph values of the buffer systems from the experiments (a,b,c,d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2


Chemistry, 22.06.2019 12:30
Avariable that is not being directly tested during an experiment should be changed varied experimented controlled
Answers: 1

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3
You know the right answer?
A0.327-g sample of azulene (c10h8) is burned in a bomb calorimeter and the temperature increases fro...
Questions

Mathematics, 18.09.2020 07:01

Mathematics, 18.09.2020 07:01

Mathematics, 18.09.2020 07:01

Mathematics, 18.09.2020 07:01

Physics, 18.09.2020 07:01

Mathematics, 18.09.2020 07:01

Mathematics, 18.09.2020 07:01

Mathematics, 18.09.2020 07:01

Mathematics, 18.09.2020 07:01

Physics, 18.09.2020 07:01

Mathematics, 18.09.2020 07:01

English, 18.09.2020 07:01

History, 18.09.2020 07:01

Mathematics, 18.09.2020 07:01

Mathematics, 18.09.2020 07:01

Mathematics, 18.09.2020 08:01

Mathematics, 18.09.2020 08:01

Mathematics, 18.09.2020 08:01

Social Studies, 18.09.2020 08:01

Geography, 18.09.2020 08:01

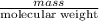
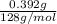
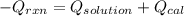
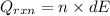
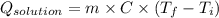
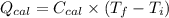
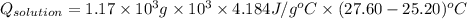
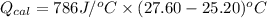
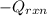 = (11748.67 + 1886.4) J
= (11748.67 + 1886.4) J
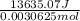
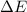 for the given combustion reaction per mole of azulene burned is 4452.26 kJ/mol.
for the given combustion reaction per mole of azulene burned is 4452.26 kJ/mol.

