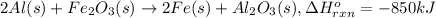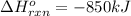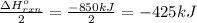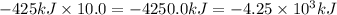Chemistry, 22.11.2019 03:31 lanashanabJHsbd1099
Calculate the enthalpy change for the thermite reaction: 2al(s)+fe2o3(s)→2fe(s)+al2o3(s), δh∘rxn=−850 kj when 10.0 mol of al undergoes the reaction with a stoichiometrically equivalent amount of fe2o3. express your answer to three significant figures and include the appropriate units.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Determine the wavelength of the light absorbed when an electron in a hydrogen atom makes a transition from an orbital in the n=3 level to an orbital in the n=7 level.
Answers: 2

Chemistry, 22.06.2019 15:30
Draw the lewis dot structure for each of the following polyatomic ions
Answers: 1

Chemistry, 22.06.2019 19:30
Anurse used a 0.02-mg/l solution of disinfection to clean a patients wound. what is the concentration of the solution expressed as a percentage?
Answers: 1

Chemistry, 22.06.2019 21:00
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1
You know the right answer?
Calculate the enthalpy change for the thermite reaction: 2al(s)+fe2o3(s)→2fe(s)+al2o3(s), δh∘rxn=−8...
Questions

Mathematics, 05.05.2020 01:28




Mathematics, 05.05.2020 01:28

Mathematics, 05.05.2020 01:28

Mathematics, 05.05.2020 01:28



Mathematics, 05.05.2020 01:28



History, 05.05.2020 01:28

Mathematics, 05.05.2020 01:28


Biology, 05.05.2020 01:28


Biology, 05.05.2020 01:28


 .
.