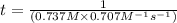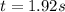Chemistry, 21.11.2019 21:31 treytonmesser
The reaction a → products was found to be second order order and have a rate constant, k, of 0.707 m-1 s-1. if the initial concentration of the reaction was 0.737 m, what is the half life for the reaction?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 08:00
Define dew point. i am writing this part to be able to ask the question
Answers: 1

Chemistry, 22.06.2019 23:00
In the reaction h2co3 (aq) + 3nh3 (aq) = 2 nh4+ (aq) + co3 2-, how many electrons are transferred?
Answers: 3

Chemistry, 23.06.2019 13:20
Which kind of weather usually forms over the northwest united states in the summer because of maritime polar air masses? 1 )fog 2)dry heat 3) heavy snow 4) heavy rain
Answers: 3

Chemistry, 23.06.2019 15:30
Sodium chloride can be made as follows: 2na + cl2 ? 2nacl i calculate the maximum amount of nacl possible if 2.3 g of sodium was reacted with excess chlorine. show all your workings.
Answers: 3
You know the right answer?
The reaction a → products was found to be second order order and have a rate constant, k, of 0.707 m...
Questions



Social Studies, 28.10.2020 08:00


Mathematics, 28.10.2020 08:00





Mathematics, 28.10.2020 08:00


Chemistry, 28.10.2020 08:00

Business, 28.10.2020 08:00

Mathematics, 28.10.2020 08:00



Mathematics, 28.10.2020 08:00

English, 28.10.2020 08:00

Biology, 28.10.2020 08:00

Chemistry, 28.10.2020 08:00

![\frac{1}{[A]_{t}}=kt+\frac{1}{[A]_{0}}](/tpl/images/0385/0648/16aaf.png)
![[A]_{t}](/tpl/images/0385/0648/c37dd.png) is concentration of A after "t" time and
is concentration of A after "t" time and ![[A]_{0}](/tpl/images/0385/0648/48818.png) is initial concentration of A
is initial concentration of A![[A]_{t}=\frac{[A]_{0}}{2}](/tpl/images/0385/0648/2b76d.png)
![[A]_{0}=0.737M](/tpl/images/0385/0648/c3ec8.png) and
and 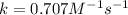
![\frac{1}{\frac{[A]_{0}}{2}}=(0.707M^{-1}s^{-1}\times t)+\frac{1}{[A]_{0}}](/tpl/images/0385/0648/7be48.png)
![\frac{1}{[A]_{0}}=0.707M^{-1}s^{-1}\times t](/tpl/images/0385/0648/fecfa.png)