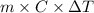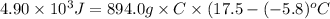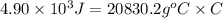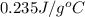Chemistry, 21.11.2019 04:31 crosales102
Achemist carefully measures the amount of heat needed to raise the temperature of a sample of a pure substance from to . the experiment shows that of heat are needed. what can the chemist report for the specific heat capacity of the substance? be sure your answer has the correct number of significant digits.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 20:00
In vapor-liquid equilibrium in a binary mixture, both components are generally present in both phases. how many degrees of freedom are there for such a system? the reaction between nitrogen and hydrogen to form ammonia occurs in the gas phase. how many degrees of freedom are there for this system? steam and coal react at high temperatures to form hydrogen, carbon monoxide, carbon dioxide, and methane. the following reactions have been suggested as being involved in the chemical transformation:
Answers: 3

Chemistry, 22.06.2019 21:40
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3

Chemistry, 23.06.2019 03:00
The size (radius) of an oxygen molecule is about 2.0 ×10−10m. make a rough estimate of the pressure at which the finite volume of the molecules should cause noticeable deviations from ideal-gas behavior at ordinary temperatures (t= 300k ). assume that deviatons would be noticeable when volume of the gas per molecule equals the volume of the molecule itself.
Answers: 3
You know the right answer?
Achemist carefully measures the amount of heat needed to raise the temperature of a sample of a pure...
Questions

English, 14.01.2021 06:00

Mathematics, 14.01.2021 06:00


English, 14.01.2021 06:00




Mathematics, 14.01.2021 06:00

German, 14.01.2021 06:00

Mathematics, 14.01.2021 06:00

History, 14.01.2021 06:00

Mathematics, 14.01.2021 06:00

Mathematics, 14.01.2021 06:00



Mathematics, 14.01.2021 06:00


Computers and Technology, 14.01.2021 06:00

Spanish, 14.01.2021 06:00

Mathematics, 14.01.2021 06:00

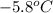 to
to 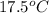 and heat needed is 4.90 kJ (or 4900 J, as 1 kJ = 1000 J).
and heat needed is 4.90 kJ (or 4900 J, as 1 kJ = 1000 J).