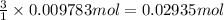The tungsten metal used for filaments in light bulbs is made by reaction of tungsten trioxide with hydrogen: wo3(s)+3h2(g)→w(s)+3h2o(g)
part a how many grams of tungsten trioxide must you start with to prepare 1.80 g of tungsten? (for wo3, mw = 231.8 amu.)
part b how many grams of hydrogen must you start with to prepare 1.80 g of tungsten?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:00
If 1.63 times 10 negative 4 of helium dissolves in 100.0g of water, what is the concentration in parts per million
Answers: 3

Chemistry, 22.06.2019 12:30
What metric units would you use to measure the thickness of a key
Answers: 3

Chemistry, 22.06.2019 12:50
What is the chemical name of the compound na2co3? use the list of polyatomic ions and the periodic table to you answer. a. sodium carbon oxide b. sodium carbonate c. sodium(ll) carbonate d. sodium oxalate
Answers: 1

Chemistry, 22.06.2019 16:00
How could a student test the effect of removing heat from a gas that is stored in a sealed container? what must occur in order for matter to change states?
Answers: 2
You know the right answer?
The tungsten metal used for filaments in light bulbs is made by reaction of tungsten trioxide with h...
Questions




















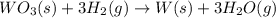
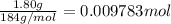
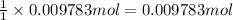 of tungsten trioxide
of tungsten trioxide