
Use the data given below to construct a born-haber cycle to determine the heat of formation of kcl. δ h°(kj) k(s) → k(g) 89 k(g) → k (g) e- 418 cl2(g) → 2 cl(g) 244 cl(g) e- → cl-(g) -349 kcl(s) → k (g) cl-(g) 717 use the data given below to construct a born-haber cycle to determine the heat of formation of kcl. h°(kj) k(s) → k(g) 89 k(g) → k (g) e- 418 cl2(g) → 2 cl(g) 244 cl(g) e- → cl-(g) -349 kcl(s) → k (g) cl-(g) 717 158 kj -1119 kj -997 kj 631 kj -437 kj

Answers: 2
Another question on Chemistry

Chemistry, 23.06.2019 06:20
Why is it that 85.48 rounded to two significant figures is 85 and not 86?
Answers: 1

Chemistry, 23.06.2019 06:30
Which of the following is true about the products formed during photosynthesis? (5 points) select one: a. they have the same mass as the mass of reactants. b. they are the same set of compounds as the reactants. c. they have more mass than the mass of reactants. d. they are chemically the same as the reactants.
Answers: 1

Chemistry, 23.06.2019 08:00
Can anyone answer these questions? ? i need it before 1: 00pm today
Answers: 1

Chemistry, 23.06.2019 09:00
A2-kg bowling ball is 1 meter off the ground on a post when it falls. just before it reaches the ground,its traveling 4.4 m/s. assuming that there is no air resistant, which statement is true a. the initial potential energy is less then the final kinetic energy b. the mechanical energy is not conserved c. the mechanical energy is conserved d. the initial potential energy is greater than the final kinetic energy
Answers: 3
You know the right answer?
Use the data given below to construct a born-haber cycle to determine the heat of formation of kcl....
Questions

Arts, 26.08.2019 00:50

Social Studies, 26.08.2019 00:50


Biology, 26.08.2019 00:50





Chemistry, 26.08.2019 00:50

Health, 26.08.2019 00:50


Biology, 26.08.2019 00:50

Biology, 26.08.2019 00:50

English, 26.08.2019 00:50


History, 26.08.2019 00:50

Biology, 26.08.2019 00:50

Computers and Technology, 26.08.2019 00:50


Mathematics, 26.08.2019 00:50

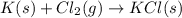
 for this reaction.
for this reaction. ,
,  = 89 kJ
= 89 kJ
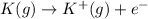 ,
,  = 418 KJ
= 418 KJ
 gas into chlorine atom
.
gas into chlorine atom
. ,
,  = 122 KJ
= 122 KJ
 ,
,  = -349 KJ
= -349 KJ
 ion and
ion and  ion formed above to get KCl
.
ion formed above to get KCl
. ,
, 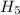 = -717 KJ
= -717 KJ



