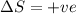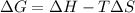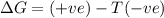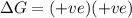Chemistry, 20.11.2019 23:31 annjetero2oy23ay
Consider the endothermic reaction: n2 (g) 2 h2 (g) → n2h4 (l) the entropy change of this reaction is and the enthalpy change is so at a very high temperature, this reaction is probably consider the endothermic reaction: n2 (g) 2 h2 (g) → n2h4 (l) the entropy change of this reaction is and the enthalpy change is so at a very high temperature, this reaction is probably unfavorable; unfavorable; nonspontaneous unfavorable; favorable; spontaneous favorable; unfavorable; spontaneous favorable; unfavorable; nonspontaneous unfavorable; unfavorable; spontaneous

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 23:40
The kw for water at 0 °c is 0.12× 10–14 m2. calculate the ph of a neutral aqueous solution at 0 °c.
Answers: 2

Chemistry, 23.06.2019 01:00
Which statement is true regarding the diagram of circle p? the sum of y and z must be 2x. the sum of y and z must be x. the difference of z and y must be 2x. the difference of z and y must be x
Answers: 1

Chemistry, 23.06.2019 02:20
Why dose heating increase the speed at which a solution dissolved in water
Answers: 1
You know the right answer?
Consider the endothermic reaction: n2 (g) 2 h2 (g) → n2h4 (l) the entropy change of this reaction i...
Questions



Chemistry, 20.03.2020 07:43

Mathematics, 20.03.2020 07:43


History, 20.03.2020 07:43

Mathematics, 20.03.2020 07:43

Mathematics, 20.03.2020 07:43

Computers and Technology, 20.03.2020 07:43


Computers and Technology, 20.03.2020 07:44






Computers and Technology, 20.03.2020 07:44



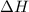 for Endothermic reaction is positive and
for Endothermic reaction is positive and 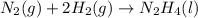
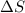 is negative as the randomness decreases when gases convert into liquid.
is negative as the randomness decreases when gases convert into liquid. 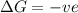 and favourable conditions are
and favourable conditions are 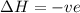 and
and