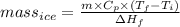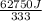Chemistry, 20.11.2019 20:31 jdsfdujfi1598
You make some iced tea by dropping 325 grams of ice into 500.0 ml of warm tea in an insulated pitcher. if the tea is initially at 30.0°c and the ice cubes are initially at 0.0°c, how many grams of ice will still be present when the contents of the pitcher reach a final temperature? the tea is mostly water, so assume that it has the same density (1.0 g/ml), molar mass, heat capacity (75.3 j/k/mol), and heat of fusion (6.01 kj/mol) as pure water. the heat capacity of ice is 37.7 j/k/mol.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 06:30
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1


Chemistry, 22.06.2019 22:30
The vapor pressure of ethanol is 1.00 × 102 mmhg at 34.90°c. what is its vapor pressure at 61.61°c? (δhvap for ethanol is 39.3 kj/mol.)
Answers: 2
You know the right answer?
You make some iced tea by dropping 325 grams of ice into 500.0 ml of warm tea in an insulated pitche...
Questions

History, 25.03.2021 17:40

Mathematics, 25.03.2021 17:40

English, 25.03.2021 17:40

Social Studies, 25.03.2021 17:40


Health, 25.03.2021 17:40

History, 25.03.2021 17:40


Mathematics, 25.03.2021 17:40

Mathematics, 25.03.2021 17:40




Mathematics, 25.03.2021 17:40

Chemistry, 25.03.2021 17:40




Mathematics, 25.03.2021 17:40

Mathematics, 25.03.2021 17:40

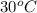 = (30 + 273) K = 303 K
= (30 + 273) K = 303 K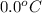 = (0 + 273) K = 273 K
= (0 + 273) K = 273 K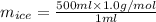
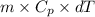
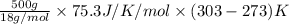
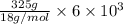
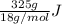 heat but we have 40774.95 J.
heat but we have 40774.95 J.