
Chemistry, 19.11.2019 20:31 harmonywilliams3064
Which of the reactions are spontaneous (favorable)? a. dhap↽−−⇀glyceraldehyde-3-phosphateδ =3.8 kj/mol b. c2h4+h2−→−−rh(i)c2h6δ=−150.97 kj/mol c. glutamate+nad++h2o⟶nh+4+α-ketogluta rate+nadh+h+δ=3.7 kcal/mol d. c6h13o9p+atp⟶c6h14o12p2+adpδ=−14.2 kj/mol e. l -malate+nad+⟶oxaloacetate+nadh+h+δ= 29.7 kj/mol

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Often on a topographic map, every fifth contour line is darkened. what is this line called? a. key b.slope c.benchmark d. index contour
Answers: 1

Chemistry, 23.06.2019 00:00
How do you determine the percent yield of a chemical reaction
Answers: 1

Chemistry, 23.06.2019 02:00
An alpha particle is: a hydrogen atom a nucleus of helium two neutrons an electron
Answers: 1

Chemistry, 23.06.2019 07:30
Which statement is actually true about the relationship between activation energy and reaction rates? low activation energy barriers result in low rates. high activation energy barriers result in low rates. low activation energy barriers result in no reaction. high activation energy barriers result in no reaction.
Answers: 2
You know the right answer?
Which of the reactions are spontaneous (favorable)? a. dhap↽−−⇀glyceraldehyde-3-phosphateδ =3.8 kj/...
Questions


Biology, 27.01.2020 20:31

Biology, 27.01.2020 20:31

Mathematics, 27.01.2020 20:31

Mathematics, 27.01.2020 20:31


Mathematics, 27.01.2020 20:31


Mathematics, 27.01.2020 20:31


Mathematics, 27.01.2020 20:31




Mathematics, 27.01.2020 20:31



Mathematics, 27.01.2020 20:31

Chemistry, 27.01.2020 20:31

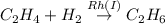 ; ΔG= −150.97 kJ/mol
; ΔG= −150.97 kJ/mol 


