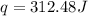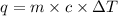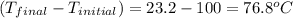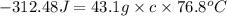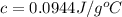You placed 43.1 g of an unknown metal at 100 °c into a coffee cup calorimeter that contained 50.0 g of water that was initially at 22.0 °c. the equilibrium temperature of mixing (t0) was determined to be 23.2 °c. the calorimeter constant was known to be 51.5 j/°c. specific heath2o = 4.184 j/g·°ca. what is the total amount of heat (j) lost by the metal? ng 1.5b. what was the specific heat (j/g·°c) of the metal? ng 1.5

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 14:00
Your answer should have the same number or significant figures as a he starting measurement. 3201 ml =
Answers: 2


Chemistry, 22.06.2019 08:00
An electron moved from shell n = 2 to shell n = 1. what most likely happened during the transition? a fraction of a photon was added. a photon of energy was absorbed. a fraction of a photon was removed. a photon of energy was released.
Answers: 1

Chemistry, 22.06.2019 17:40
If 3 moles of a compound use 24 j of energy in a reaction, what is the a hreaction in j/mol?
Answers: 1
You know the right answer?
You placed 43.1 g of an unknown metal at 100 °c into a coffee cup calorimeter that contained 50.0 g...
Questions

Health, 22.07.2019 13:00



History, 22.07.2019 13:00



Biology, 22.07.2019 13:00

Spanish, 22.07.2019 13:00




History, 22.07.2019 13:00

Biology, 22.07.2019 13:00

Mathematics, 22.07.2019 13:00

Mathematics, 22.07.2019 13:00


Mathematics, 22.07.2019 13:00

Health, 22.07.2019 13:00

Spanish, 22.07.2019 13:00

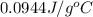
![q=[q_1+q_2]](/tpl/images/0381/4030/341bc.png)
![q=[c_1\times \Delta T+m_2\times c_2\times \Delta T]](/tpl/images/0381/4030/1d50b.png)
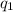 = heat absorbed by the calorimeter
= heat absorbed by the calorimeter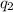 = heat absorbed by the water
= heat absorbed by the water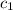 = specific heat of calorimeter =
= specific heat of calorimeter = 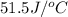
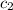 = specific heat of water =
= specific heat of water = 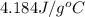
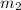 = mass of water = 50.0 g
= mass of water = 50.0 g = change in temperature =
= change in temperature = 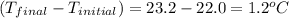
![q=[(51.5J/^oC\times 1.2^oC)+(50.0g\times 4.184J/g^oC\times 1.2^oC)]](/tpl/images/0381/4030/aca3d.png)