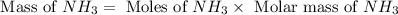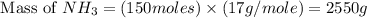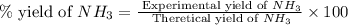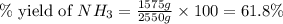Chemistry, 19.11.2019 06:31 jlbradley429
The reaction n2 + 3 h2 → 2 nh3 is used to produce ammonia. when 450. g of hydrogen was reacted with nitrogen, 1575 g of ammonia were produced. what is the percent yield of this reaction?
30.8%
61.8%
20.7%
41.5%
more information is needed to solve this problem.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:20
Calcium hydride (cah2) reacts with water to form hydrogen gas: cah2(s) + 2h2o(l) → ca(oh)2(aq) + 2h2(g) how many grams of cah2 are needed to generate 45.0 l of h2 gas at a pressure of 0.995 atm and a temperature of 32 °c?
Answers: 2

Chemistry, 22.06.2019 00:30
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1

Chemistry, 22.06.2019 19:00
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1

Chemistry, 22.06.2019 23:00
What is the average rate of the reaction between 10 and 20 s?
Answers: 1
You know the right answer?
The reaction n2 + 3 h2 → 2 nh3 is used to produce ammonia. when 450. g of hydrogen was reacted with...
Questions

Chemistry, 09.03.2021 17:10

Mathematics, 09.03.2021 17:10

Mathematics, 09.03.2021 17:10

History, 09.03.2021 17:10


Physics, 09.03.2021 17:10

Mathematics, 09.03.2021 17:10

Mathematics, 09.03.2021 17:10

Health, 09.03.2021 17:10

Advanced Placement (AP), 09.03.2021 17:10

Mathematics, 09.03.2021 17:10

Mathematics, 09.03.2021 17:10

Mathematics, 09.03.2021 17:10


Mathematics, 09.03.2021 17:10

Mathematics, 09.03.2021 17:10

Mathematics, 09.03.2021 17:10


Mathematics, 09.03.2021 17:10

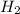 = 450 g
= 450 g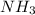 = 17 g/mole
= 17 g/mole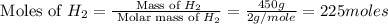
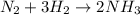
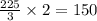 moles of
moles of