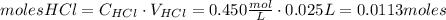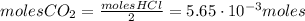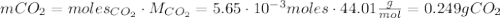Chemistry, 19.11.2019 06:31 caromaybelline71
Calcium carbonate (caco3) reacts with stomach acid (hcl, hydrochloric acid) according to the following equation: caco3(s) 2hcl(aq)⟶co2(g) h2o(l) cacl2(aq) a typical antacid contains caco3. if such an antacid is added to 25.0 ml of a solution that is 0.450 m in hcl, how many grams of co2 gas are produced?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:00
At 450 mm hg a gas has a volume of 760 l, what is its volume at standard pressure
Answers: 2

Chemistry, 22.06.2019 14:50
How are evaporation and sublimation similar? a both involve the formation of a gas. b both release energy to the surroundings. c both take place throughout a solid. d both take place at the surface of a liquid.
Answers: 1

Chemistry, 22.06.2019 17:30
Upon decomposition, one sample of magnesium fluoride produced 1.65 kg of magnesium and 2.56 kg of fluorine. a second sample produced 1.32 kg of magnesium. part a how much fluorine (in grams) did the second sample produce?
Answers: 2

Chemistry, 22.06.2019 18:00
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3
You know the right answer?
Calcium carbonate (caco3) reacts with stomach acid (hcl, hydrochloric acid) according to the followi...
Questions


History, 25.06.2019 11:00

Mathematics, 25.06.2019 11:00

English, 25.06.2019 11:00


Mathematics, 25.06.2019 11:00


Business, 25.06.2019 11:00





Advanced Placement (AP), 25.06.2019 11:00




Biology, 25.06.2019 11:00

Social Studies, 25.06.2019 11:00

Computers and Technology, 25.06.2019 11:00