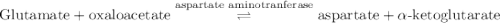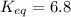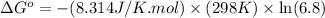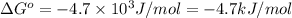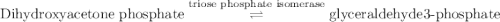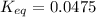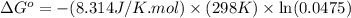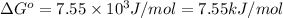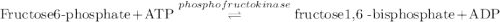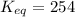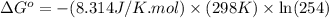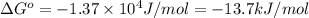Chemistry, 19.11.2019 03:31 joseenrique02
Calculation of δg′° from an equilibrium constant calculate the standard free-energy change for each of the following metabolically important enzyme-catalyzed reactions, using the equilibrium constants given for the reactions at 25 °c and ph 7.0. ( a ) glutamate + oxaloacetate aspartate aminotranferase ⇌ aspartate + α -ketoglutarate k ′ eq = 6.8 ( b ) dihydroxyacetone phosphate triose phosphate isomerase ⇌ glyceraldehyde 3 -phosphate k ′ eq = 0.0475 ( c ) fructose 6 -phosphate + atp phosphofructokinase ⇌ fructose 1 , 6 -bisphosphate + adp k ′ eq = 254

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Imagine that you’re getting ready to move to a new city. when people move, they are influenced by push factors and pull factors, and you have many reasons for your move. which of the following factors is an example of a pull factor? a. wanting to move because you’ve found a great new school somewhere new b. needing to move because there are not enough resources in your old hometown c. being forced to move because your old home is gone d. having to move because there are no jobs in your current hometown
Answers: 1

Chemistry, 21.06.2019 22:50
2. you__turn left on a red light if you are in the left-most lane of a one-way street, you're turning into the left-most lane of a one-way street, and no nearby sign prohibits the turn.
Answers: 2

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3

Chemistry, 22.06.2019 08:40
Write the formula for the following chemicals. 7. e. trinitrogen tetraoxide a calcium phosphate f. magnesium acetate b. potassium sulfide g nickel(iii) cyanide c carbon dioxide h. silver sulfate d. cobalt(ii) chloride
Answers: 1
You know the right answer?
Calculation of δg′° from an equilibrium constant calculate the standard free-energy change for each...
Questions










Mathematics, 23.08.2021 17:00

Medicine, 23.08.2021 17:00

Chemistry, 23.08.2021 17:00




Mathematics, 23.08.2021 17:00

Business, 23.08.2021 17:00

Social Studies, 23.08.2021 17:00


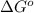 for the reaction is -4.7 kJ/mol
for the reaction is -4.7 kJ/mol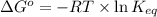
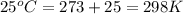
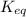 = equilibrium constant
= equilibrium constant