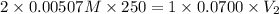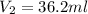Chemistry, 19.11.2019 01:31 fluffylove83
Achemistry student weighs out of sulfurous acid , a diprotic acid, into a volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with solution. a. calculate the volume of solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 12:30
4nh3+5o2--> 4no+6h20what is the total number of moles of h2o produced when 12 mole of nh3 is completely consumed?
Answers: 3

Chemistry, 21.06.2019 22:00
To save time, you can approximate the initial mass of the solid to the nearest ±1 g. for example, if you are asked to add 14.3 g of copper, add between 13 g and 15 g. which of the following sets include two samples with an equal density? which all that apply below 15.4 g gold and 18.7 g silver 15.2 g copper and 50.0 g copper 20.2 g silver and 20.2 g copper 11.2 g gold and 14.9 g gold
Answers: 1

Chemistry, 21.06.2019 23:00
A100-watt light bulb radiates energy at a rate of 100 j/s. (the watt, a unit of power or energy over time, is defined as 1 j/s.) if all of the light emitted has a wavelength of 525 nm , how many photons are emitted per second?
Answers: 1

You know the right answer?
Achemistry student weighs out of sulfurous acid , a diprotic acid, into a volumetric flask and dilut...
Questions

French, 17.03.2021 23:50

History, 17.03.2021 23:50


Mathematics, 17.03.2021 23:50

Chemistry, 17.03.2021 23:50


French, 17.03.2021 23:50



Computers and Technology, 17.03.2021 23:50

Mathematics, 17.03.2021 23:50




Mathematics, 17.03.2021 23:50

Mathematics, 17.03.2021 23:50



Mathematics, 17.03.2021 23:50

Mathematics, 17.03.2021 23:50

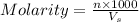
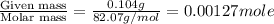
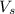 = volume of solution in ml
= volume of solution in ml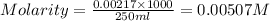
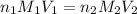
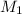 = molarity of
= molarity of 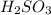 solution = 0.00507 M
solution = 0.00507 M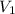 = volume of
= volume of 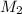 = molarity of
= molarity of 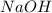 solution = 0.0700 M
solution = 0.0700 M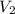 = volume of
= volume of 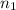 = valency of
= valency of 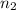 = valency of
= valency of