
Chemistry, 16.11.2019 05:31 andregijoe41
What is the molarity of zncl2 that forms when 25.0 g of zinc completely reacts with cucl2 according to the following reaction? assume a final volume of 285 ml .
zn(s)+cucl2(aq)→zncl2(aq)+cu(s)

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 01:30
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 08:30
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1

Chemistry, 23.06.2019 02:00
An alpha particle is: a hydrogen atom a nucleus of helium two neutrons an electron
Answers: 1
You know the right answer?
What is the molarity of zncl2 that forms when 25.0 g of zinc completely reacts with cucl2 according...
Questions

Mathematics, 24.06.2019 20:30

Chemistry, 24.06.2019 20:30

Chemistry, 24.06.2019 20:30

History, 24.06.2019 20:30

French, 24.06.2019 20:30



Mathematics, 24.06.2019 20:30




Mathematics, 24.06.2019 20:30



History, 24.06.2019 20:30


History, 24.06.2019 20:30

Mathematics, 24.06.2019 20:30


History, 24.06.2019 20:30

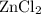 is 1.364 M
is 1.364 M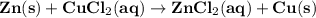
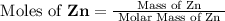 , molar mass of Zn = 65.38 g/mol
, molar mass of Zn = 65.38 g/mol 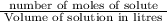
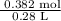 = 1.364 M.
= 1.364 M.

