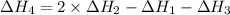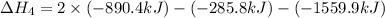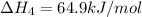1) the heat of combustion for the gases hydrogen, methane and ethane are −285.8, −890.4 and −1559.9 kj/mol respectively at 298k.
equation 1 h2 + 1⁄2o2 > h2o δh = −285.8 kj
equation 2 ch4 + 2o2 > co2 + 2h2o δh = −890.4 kj
equation 3 c2h6 + 7⁄2o2 > 2co2 + 3h2o δh = −1559.9 kj
use the above equations to calculate (at the same temperature) the heat of reaction for the following reaction:
2ch4(g) > c2h6(g) + h2(g)
solution:

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Sylvanite is a mineral that contains 28.0% gold by mass. how much sylvanite would you need to dig up to obtain 77.0 g of gold? explain how you got your answer and the steps you took. you
Answers: 3

Chemistry, 21.06.2019 22:30
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3

Chemistry, 22.06.2019 08:30
Joan writes four numbers on the board in standard form, and then she writes their scientific notation
Answers: 1

Chemistry, 22.06.2019 16:50
Which element is least likely to undergo a chemical reaction
Answers: 3
You know the right answer?
1) the heat of combustion for the gases hydrogen, methane and ethane are −285.8, −890.4 and −1559.9...
Questions

Mathematics, 10.04.2020 01:28



Mathematics, 10.04.2020 01:28

Biology, 10.04.2020 01:28

History, 10.04.2020 01:28


History, 10.04.2020 01:28

History, 10.04.2020 01:28






Biology, 10.04.2020 01:28


Chemistry, 10.04.2020 01:28



Mathematics, 10.04.2020 01:28

 ..[1]
..[1]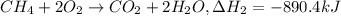 ..[2]
..[2]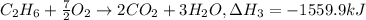 ..[3]
..[3]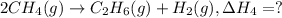 ..[4]
..[4]