
Chemistry, 10.11.2019 05:31 battlemarshmell
Aqueous sulfuric acid reacts with solid sodium hydroxide to produce aqueous sodium sulfate and liquid water . if of sodium sulfate is produced from the reaction of of sulfuric acid and of sodium hydroxide, calculate the percent yield of sodium sulfate. be sure your answer has the correct number of significant digits in it.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

Chemistry, 22.06.2019 11:30
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2

Chemistry, 22.06.2019 17:50
You exhale co2 which is produced during cellular respiration. co2 combines with the water in your blood's plasma to make up one half of the body's most important buffer pair, carbonic acid. the more physical activity you engage in, the more co2 your body is producing. you can see this by putting some of the cabbage indicator in a glass and then blowing bubbles into it through a straw. can you see a change in the color of the indicator?
Answers: 2

Chemistry, 23.06.2019 00:00
How many moles of water are created if 3 moles of hydrogen react completely with excess oxygen?
Answers: 3
You know the right answer?
Aqueous sulfuric acid reacts with solid sodium hydroxide to produce aqueous sodium sulfate and liqui...
Questions

English, 17.01.2021 22:40

Mathematics, 17.01.2021 22:40

Mathematics, 17.01.2021 22:40

Mathematics, 17.01.2021 22:50

History, 17.01.2021 22:50


Physics, 17.01.2021 22:50


Social Studies, 17.01.2021 22:50



Business, 17.01.2021 22:50


History, 17.01.2021 22:50

Mathematics, 17.01.2021 22:50

Mathematics, 17.01.2021 22:50

Mathematics, 17.01.2021 22:50


Geography, 17.01.2021 22:50


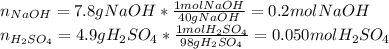
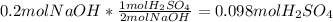
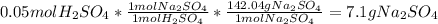
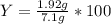
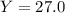 %
%

