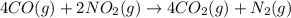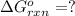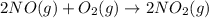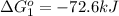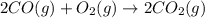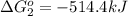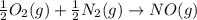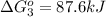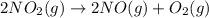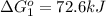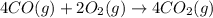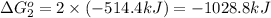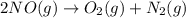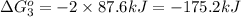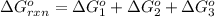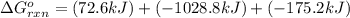Chemistry, 10.11.2019 01:31 TheOriginal2x
Calculate δg∘rxn for the following reaction: 4co(g)+2no2(g)→4co2(g)+n2(g).use the following reactions and given δg∘rxn values: a) 2no(g)+o2(g)→2no2(g), δg∘rxn= - 72.6 kjb) 2co(g)+o2(g)→2co2(g), δg∘rxn= - 514.4 kjc) 12o2(g)+12n2(g)→no(g), δg∘rxn= 87.6 kj

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:40
Which statement is usually true about the relationship between activation energy and reaction rates? low activation energy barriers result in low rates. high activation energy barriers result in low rates. low activation energy barriers result in no reaction. high activation energy barriers result in no reaction.
Answers: 3

Chemistry, 22.06.2019 08:40
Which statement can best be concluded from the ideal gas law?
Answers: 2

Chemistry, 22.06.2019 13:30
Table sugar completely dissolved in water is an example of a?
Answers: 1

Chemistry, 22.06.2019 19:10
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2
You know the right answer?
Calculate δg∘rxn for the following reaction: 4co(g)+2no2(g)→4co2(g)+n2(g).use the following reaction...
Questions

Mathematics, 23.03.2021 19:50


Chemistry, 23.03.2021 19:50




Mathematics, 23.03.2021 19:50

Mathematics, 23.03.2021 19:50

Mathematics, 23.03.2021 19:50


Mathematics, 23.03.2021 19:50



Mathematics, 23.03.2021 19:50



Mathematics, 23.03.2021 19:50



Mathematics, 23.03.2021 19:50

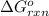 for the reaction is -1131.4 kJ
for the reaction is -1131.4 kJ