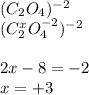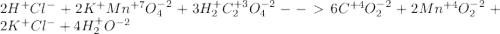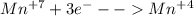Chemistry, 08.11.2019 01:31 JamesLachoneus
In an ion, the sum of the oxidation states is equal to the overall ionic charge. note that the sign of the oxidation states and the number of atoms associated with each oxidation state must be considered. in oh-, for example, the oxygen atom has an oxidation state of 2 and the hydrogen atom has an oxidation state of +1, for a total of ( -2 ) + ( +1 ) = -1
(i) what is the oxidation state of each individual carbon atom in c2o4 2^-
express the oxidation state numerically (e. g., +1).
(ii) which element is reduced in this reaction?
2hcl+ 2kmno4 + 3h2c2o4 ? 6co2 +2mno2 + 2kcl + 4h2o

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 03:30
If you have 5.25 grams of methane (ch4), how many grams of co2 will you produce ?
Answers: 1

Chemistry, 22.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 1

Chemistry, 22.06.2019 05:30
You are making a solution of calcium chloride dissolved in water. you add solid, stir, and it dissolves. you add just a spatula tip full, stir, and the solid does not dissolve. how could you describe the solutions before and after adding the spatula tip amount
Answers: 1
You know the right answer?
In an ion, the sum of the oxidation states is equal to the overall ionic charge. note that the sign...
Questions

Chemistry, 30.11.2019 05:31

Social Studies, 30.11.2019 05:31


Mathematics, 30.11.2019 05:31





Mathematics, 30.11.2019 05:31






Spanish, 30.11.2019 05:31

Chemistry, 30.11.2019 05:31


Social Studies, 30.11.2019 05:31

History, 30.11.2019 05:31