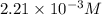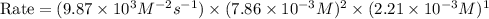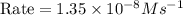Chemistry, 08.11.2019 00:31 giavanleer14
2no + o22 no2 is second order in no and first order in o2. complete the rate law for this reaction in the box below. use the form , where '1' is understood for m, n (don't enter 1) and concentrations taken to the zero power do not appear. rate = in an experiment to determine the rate law, the rate constant was determined to be 9.87×103 m-2s-1. using this value for the rate constant, the rate of the reaction when [no] = 7.86×10-3 m and [o2] = 2.21×10-3 m would be ms-1.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 1


Chemistry, 23.06.2019 03:50
Show how to convert the temperature 84.7° c to kelvin. include all steps and label the final answer.
Answers: 1
You know the right answer?
2no + o22 no2 is second order in no and first order in o2. complete the rate law for this reaction i...
Questions



Physics, 19.11.2020 04:50

Mathematics, 19.11.2020 04:50


Mathematics, 19.11.2020 04:50

Mathematics, 19.11.2020 04:50

Mathematics, 19.11.2020 04:50


Mathematics, 19.11.2020 04:50

Mathematics, 19.11.2020 04:50


Arts, 19.11.2020 04:50

Biology, 19.11.2020 04:50



English, 19.11.2020 04:50

Mathematics, 19.11.2020 04:50


Mathematics, 19.11.2020 04:50

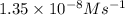
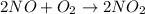
![\text{Rate}=k[NO]^a[O_2]^b](/tpl/images/0364/5919/1903b.png)
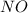 = 2
= 2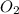 = 1
= 1![\text{Rate}=k[NO]^2[O_2]^1](/tpl/images/0364/5919/3eb46.png)
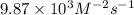
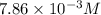
![[O_2]](/tpl/images/0364/5919/b0db0.png) = concentration of
= concentration of