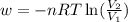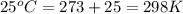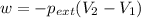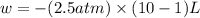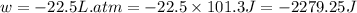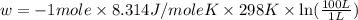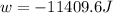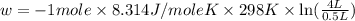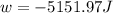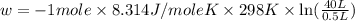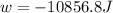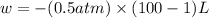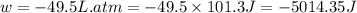Order the follow processes from (1) the least work done by the system to (5) the most work done by one mole of an ideal gas at 25°c. 1. an isothermal expansion from 1 l to 10 l at an external pressure of 2.5 atm. 2. a free isothermal expansion from 1 l to 100 l. 3. a reversible isothermal expansion from 0.5 l to 4 l. 4. a reversible isothermal expansion from 0.5 l to 40 l. 5. an isothermal expansion from 1 l to 100 l at an external pressure of 0.5 atm.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 12:40
Numbers with a end value of 4 or lower are rounded up, while with an end value of 5 or higher are rounded down. true or false?
Answers: 1

Chemistry, 21.06.2019 20:30
14. complete and balance the equations for the single displacement reactions. a. zn + pb(no3)2 -> b. al + niso4 -> 15. complete and balance the equations for the double displacement reactions. a. agno3(aq) + nacl(aq) -> b. mg(no3)2(aq) + koh(aq) -> 16. complete and balance the equations for the combustion reactions. a. __ ch4 + o2 -> b. __ c3h6 + o2 -> c. + o2 ->
Answers: 2


You know the right answer?
Order the follow processes from (1) the least work done by the system to (5) the most work done by o...
Questions

Mathematics, 20.08.2019 17:30

History, 20.08.2019 17:30

Mathematics, 20.08.2019 17:30


Biology, 20.08.2019 17:30




History, 20.08.2019 17:30




Mathematics, 20.08.2019 17:30

English, 20.08.2019 17:30


Mathematics, 20.08.2019 17:30

Mathematics, 20.08.2019 17:30

Chemistry, 20.08.2019 17:30


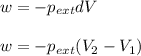
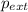 = external pressure
= external pressure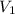 = initial volume of gas
= initial volume of gas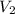 = final volume of gas
= final volume of gas