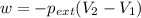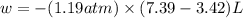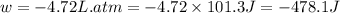Asample of gas in a cylinder of volume 3.42 l at 298 k and 2.57 atm expands to 7.39 l by two different pathways. path a is an isothermal, reversible expansion. path b has two steps. in the fi rst step, the gas is cooled at constant volume to 1.19 atm. in the second step, the gas is heated and allowed to expand against a constant external pressure of 1.19 atm until the fi nal volume is 7.39 l. calculate the work for each path.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Drag each label to the correct location on the chart. classify each reaction as endothermic or exothermic.
Answers: 1


Chemistry, 22.06.2019 17:30
Aroller coaster is traveling at 13 mi./s when you purchase a hill that is 400 m long and down the hill exonerate at 4.0 m/s squared what is the final velocity of the posterior found your answer to the nearest number
Answers: 1

You know the right answer?
Asample of gas in a cylinder of volume 3.42 l at 298 k and 2.57 atm expands to 7.39 l by two differe...
Questions




Computers and Technology, 22.07.2019 11:00

Biology, 22.07.2019 11:00

Chemistry, 22.07.2019 11:00

Chemistry, 22.07.2019 11:00












Social Studies, 22.07.2019 11:00

Geography, 22.07.2019 11:00

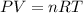
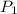 = initial pressure of gas = 2.57 atm
= initial pressure of gas = 2.57 atm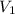 = initial volume of gas = 3.42 L
= initial volume of gas = 3.42 L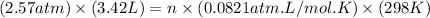
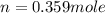
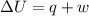
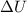 = internal energy
= internal energy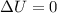
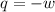
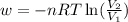
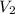 = final volume of gas = 7.39 L
= final volume of gas = 7.39 L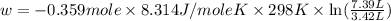
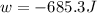
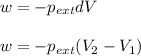
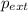 = external pressure = 1.19 atm
= external pressure = 1.19 atm