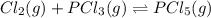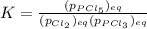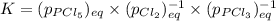Which equilibrium constant expression(s) are for the following reaction. (x stands for the mole fraction of the indicated substance in its phase, which we have so far assumed equal to unity (one) for essentially pure phases.)
cl2(g) + pcl3(g)↔pcl5(g))
choose from the list below and enter the letters alphabetical order. (e. g. ah)
(a) (pcl2)eq
(b) (ppcl3)eq
(c) (ppcl5)eq
(d) (pcl2)eq-1
(e) (ppcl3)eq-1
(f) (ppcl5)eq-1
(g) (pcl2)eq2
(h) (ppcl3)eq3
(i) (ppcl5)eq5
(j) (pcl2)eq-2
(k) (ppcl3)eq-3
(l) (ppcl5)eq-5

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:20
What is the strongest intermolecular force between an nacl unit and an h2o molecule together in a solution? covalent bonding dipole-dipole force hydrogen bonding ion-dipole force
Answers: 1


Chemistry, 22.06.2019 08:50
If two atoms are bonded to a central atom with no lone pairs,how will they be arranged
Answers: 3

Chemistry, 22.06.2019 13:00
The molality of calcium chloride (cacl2) in an aqueous solution is 2.46 m. what is mole fraction of the solute?
Answers: 3
You know the right answer?
Which equilibrium constant expression(s) are for the following reaction. (x stands for the mole frac...
Questions














Computers and Technology, 29.11.2019 03:31






 .
.