
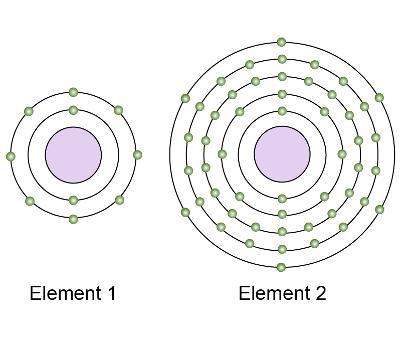
These models show the electron structures of two different nonmetal elements.
which element is likely more reactive, and why?
a.) element 1 is more reactive because it has fewer electron shells and is toward the top of its group on the periodic table.
b.) element 1 is more reactive because it has more electrons in its valence shell and is farther to the right on the periodic table.
c.) element 2 is more reactive because it does not have a valence shell close to the nucleus, so it will attract electrons.
d.) element 2 is more reactive because it does not have a full valence shell, so it will attract electrons.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:30
Determine the number o moles of ions/atoms/particle in the following: 2.50 miles of k2s (let me know how to do)
Answers: 1

Chemistry, 22.06.2019 02:30
The is a particle with one unit of positive charge a. proton b. positron c. electron d. nucleus awnser quick it is a important science test!
Answers: 2

Chemistry, 22.06.2019 09:20
What will most likely happen when two bromine atoms bond together?
Answers: 3

Chemistry, 22.06.2019 23:00
What is the energy in joules of a mole of photons associated with visible light of wavelength 486 nm?
Answers: 3
You know the right answer?
These models show the electron structures of two different nonmetal elements.
which elem...
which elem...
Questions






Geography, 05.09.2020 14:01


Computers and Technology, 05.09.2020 14:01






Geography, 05.09.2020 14:01



History, 05.09.2020 14:01





