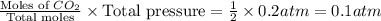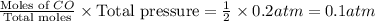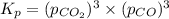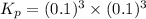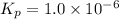When heated, lanthanum(iii) oxalate decomposes as follows: la2(c2o4)3(s) < ===> la2o3(s) + 3 co2(g) + 3 co(g) starting with just the oxalate in a 10.0 l flask, at equilibrium the total pressure observed is 0.200 atm. what is the value of kp for the equilibrium? (dalton’s law of partial pressure! )

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:30
What is the formula for the molecular compound nitrogen monoxide
Answers: 1

Chemistry, 22.06.2019 19:00
How many moles of cu are needed to react with 5.8 moles of agno3? cu + 2 agno3 → cu(no3)2 + 2 ag
Answers: 3

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

Chemistry, 22.06.2019 22:00
Does the number of ions in solution increase, decrease, or remain constant? it continuously decreases. it continuously increases. it decreases at first, then increases. it increases at first, then decreases.
Answers: 3
You know the right answer?
When heated, lanthanum(iii) oxalate decomposes as follows: la2(c2o4)3(s) < ===> la2o3(s) + 3...
Questions

Mathematics, 02.07.2019 06:00

Mathematics, 02.07.2019 06:00

Mathematics, 02.07.2019 06:00


English, 02.07.2019 06:00

History, 02.07.2019 06:00

History, 02.07.2019 06:00


History, 02.07.2019 06:00

English, 02.07.2019 06:00

Mathematics, 02.07.2019 06:00




Biology, 02.07.2019 06:00


Mathematics, 02.07.2019 06:00

Biology, 02.07.2019 06:00

Mathematics, 02.07.2019 06:00

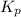 for the equilibrium is
for the equilibrium is 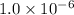
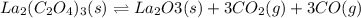
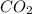 and
and 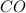 .
.