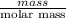Chemistry, 28.10.2019 21:31 alarconanais07
The following procedure provides a crude method of determining the molar mass of a volatile liquid. a liquid of mass 0.0224 g is introduced into a syringe and the end is capped (sealed). the syringe is transferred to a temperature bath maintained at 49.9 oc, and the liquid vaporizes. as the liquid vaporizes the plunger is pushed out. at equilibrium, the plunger reads 6.56 ml of gas. atmospheric pressure is 740. mmhg. what is the approximate molar mass of the compound (in g/mol)?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 14:00
Diamond, graphite, and fullerenes share what property? a. they are all made of carbon (c) bonded to a metal. b. their shape. c. they are all made of carbon (c). d. they are all good conductors.
Answers: 1

Chemistry, 21.06.2019 22:30
What are the charges of the subatomic particles by choosing the answer from the drop down menu. protons have a (+1,0,or-1). (protons,neutrons,electrons) have a 0 charge. 3.) electrons have a (+1,0,-1)
Answers: 2

Chemistry, 22.06.2019 02:00
In which of these cases are the two wave points considered to be in phase with each other?
Answers: 1

Chemistry, 22.06.2019 13:00
These questions are based on the attached photo. the experiment is about burning magnesium metal with oxygen. 1. write the balanced chemical equation for the reaction you are performing. 2. calculate the mass of magnesium metal used in each trial. o trial 1: o trial 2: 3. calculate the actual yield of magnesium oxide for each trial. o trial 1: o trial 2: 4. magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. o trial 1: o trial 2: 5. determine the percent yield of mgo for your experiment for each trial. o trial 1: o trial 2: 6. determine the average percent yield of mgo for the two trials. your company currently uses a process with a similar cost of materials that has an average percent yield of 91 percent. if the average percent yield of this process is higher than that, this could save the company money. what is your recommendation to the company? support your recommendation using your data, calculations, and understanding of stoichiometry gathered from this lab.
Answers: 1
You know the right answer?
The following procedure provides a crude method of determining the molar mass of a volatile liquid....
Questions



German, 18.11.2020 01:00


Mathematics, 18.11.2020 01:00




English, 18.11.2020 01:00

Health, 18.11.2020 01:00



Mathematics, 18.11.2020 01:00


Mathematics, 18.11.2020 01:00

Computers and Technology, 18.11.2020 01:00

History, 18.11.2020 01:00


Mathematics, 18.11.2020 01:00


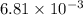 L
L


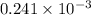 moles
moles