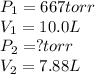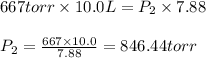Chemistry, 28.10.2019 13:31 floreschachi8230
Fixed amount of gas at 25.0 °c occupies a volume of 10.0 l when the pressure is 667 torr. use boyle's law to calculate the pressure (torr) when the volume is reduced to 7.88 l at a constant temperature of 25.0°c

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:50
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3 -> a2cr(no3)3 + a3ag2so4
Answers: 1

Chemistry, 22.06.2019 08:00
Match the mixture with the substance// i really need on this guys (it’s a pic btw)
Answers: 1

Chemistry, 22.06.2019 19:00
Structure of the atoms: discovery of the nucleus in 1909i need answering all of these questions
Answers: 3

Chemistry, 22.06.2019 23:20
In medium-sized stars such as the sun, nuclear fusion almost always means the fusing of nuclei to form , but larger stars can produce elements as heavy as
Answers: 2
You know the right answer?
Fixed amount of gas at 25.0 °c occupies a volume of 10.0 l when the pressure is 667 torr. use boyle'...
Questions

History, 19.10.2019 21:30

Health, 19.10.2019 21:30





Mathematics, 19.10.2019 21:30




Biology, 19.10.2019 21:30


Chemistry, 19.10.2019 21:30

Mathematics, 19.10.2019 21:30

Mathematics, 19.10.2019 21:30

Mathematics, 19.10.2019 21:30


Health, 19.10.2019 21:30


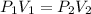
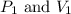 are initial pressure and volume of the gas.
are initial pressure and volume of the gas.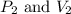 are final pressure and volume of the gas
are final pressure and volume of the gas