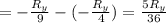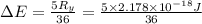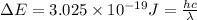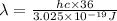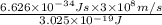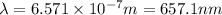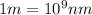The energy e of the electron in a hydrogen atom can be calculated from the bohr formula:
e= -ry/n^2
in this equation ry stands for the rydberg energy, and n stands for the principal quantum number of the orbital that holds the electron.
calculate the wavelength of the line in the absorption line spectrum of hydrogen caused by the transition of the electron from an orbital with n=2 to an orbital with n=3.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 15:00
Describe what happens to the molecules as water goes from ice to liquid to vapor. be sure to explain what happens to the temperature during the phase changes.
Answers: 2


Chemistry, 22.06.2019 16:40
The diagram below shows the movement of particles. what does this piece of evidence best support? the collision theory the maxwell-boltzmann distribution the effect of pressure on reaction rates the effect of temperature on reaction rates
Answers: 3

Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2
You know the right answer?
The energy e of the electron in a hydrogen atom can be calculated from the bohr formula:
Questions

English, 21.11.2020 02:00

Biology, 21.11.2020 02:00

Mathematics, 21.11.2020 02:00

Mathematics, 21.11.2020 02:00

French, 21.11.2020 02:00

Health, 21.11.2020 02:00

Mathematics, 21.11.2020 02:00

Physics, 21.11.2020 02:00


Mathematics, 21.11.2020 02:00




English, 21.11.2020 02:00


History, 21.11.2020 02:00

Computers and Technology, 21.11.2020 02:00

History, 21.11.2020 02:00

English, 21.11.2020 02:00

Mathematics, 21.11.2020 02:00

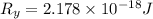
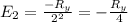
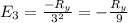
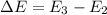 :
: