
Chemistry, 24.10.2019 03:00 morganlynn18
You have prepared a buffer by combining 5.00 ml 0.010 m ha (weak acid) and 3.00 ml 0.010 m naa (provides a -, the conjugate base of ha). pk a for ha is 6.23. how many moles of a - will be present in the mixture after addition of 0.20 ml 0.010 m naoh? (suggestion: start by calculating the moles of everything in the mixture.) enter the numerical value, without units, to at least 2 significant figures. you may use e-format scientific notation, e. g., 1.23x10 -4 would be entered as 1.23e-4. (note that the acid and base do not add up to 20.00 ml -- water must be added. that does not affect how you answer the question.)

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:30
Imagine that you own a property that is exactly 2.2 acres large. you want to sell your property, but your realtor tells you that you cannot sell your land by the acre. in order to sell your land you need to determine the area you own in units of square meters? given that there are 1.6 kilometers in 1 mile and 640 acres in 1 square mile, what is the area of land that you own in square meters square meters?
Answers: 2

Chemistry, 22.06.2019 03:10
The covalent compound acetylene, which is the fuel of the oxyacetylene torch used by welders, has the molecular formula c2h2. the covalent compound benzene, a commercial solvent, has the molecular formula c6h6 each of these covalent compounds contains carbon and hydrogen atoms in a one-to-one ratio. would it be correct to write the chemical formulas of each as ch? explain.
Answers: 1

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 12:00
Ageochemist examines a piece of metal that he found in the soil. he performs tests to identify the metal from its density, electrical conductivity, and melting point. which statement best describes his investigation? a. he is determining physical properties that are sufficient to identify the metal.b. he is determining chemical properties that are sufficient to identify the metal.c. he is determining physical properties that are insufficient to identify the metal.d. he is determining chemical properties that are insufficient to identify the metal.
Answers: 3
You know the right answer?
You have prepared a buffer by combining 5.00 ml 0.010 m ha (weak acid) and 3.00 ml 0.010 m naa (prov...
Questions


Biology, 15.04.2020 20:17



Social Studies, 15.04.2020 20:17


Computers and Technology, 15.04.2020 20:17


Biology, 15.04.2020 20:17







English, 15.04.2020 20:17


Social Studies, 15.04.2020 20:18


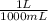 = 5x10⁻³ L of HA
= 5x10⁻³ L of HA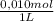 = 5x10⁻⁵ mol of HA
= 5x10⁻⁵ mol of HA

